The fight against tuberculosis (TB) remains one of the most significant challenges in global public health. Often perceived as a relic of the past, TB has surged back to the forefront of medical urgency, surpassing COVID-19 to once again become the world’s leading cause of death from a single infectious agent. In 2024 alone, an estimated 10.7 million people fell ill with the disease, and approximately 1.23 million lost their lives; a tragic figure given that TB is both preventable and curable. The danger of the disease is compounded by its airborne nature and its ability to thrive where poverty, malnutrition, and overcrowded housing persist. Furthermore, the rise of multidrug-resistant TB (MDR-TB) represents a public health crisis and a threat to global health security, as only about two in five people with drug-resistant strains managed to access treatment in the last year.
In countries like Iraq, where healthcare infrastructure must adapt to unique regional pressures, the need for specialized, robust management systems is paramount. It is within this critical context that eSITE, a Baghdad-based IT solutions provider established in 2015, partnered with the International Organization for Migration (IOM) and the National Tuberculosis Institute to design and implement Iraq’s National Tuberculosis Programme (NTP) System.
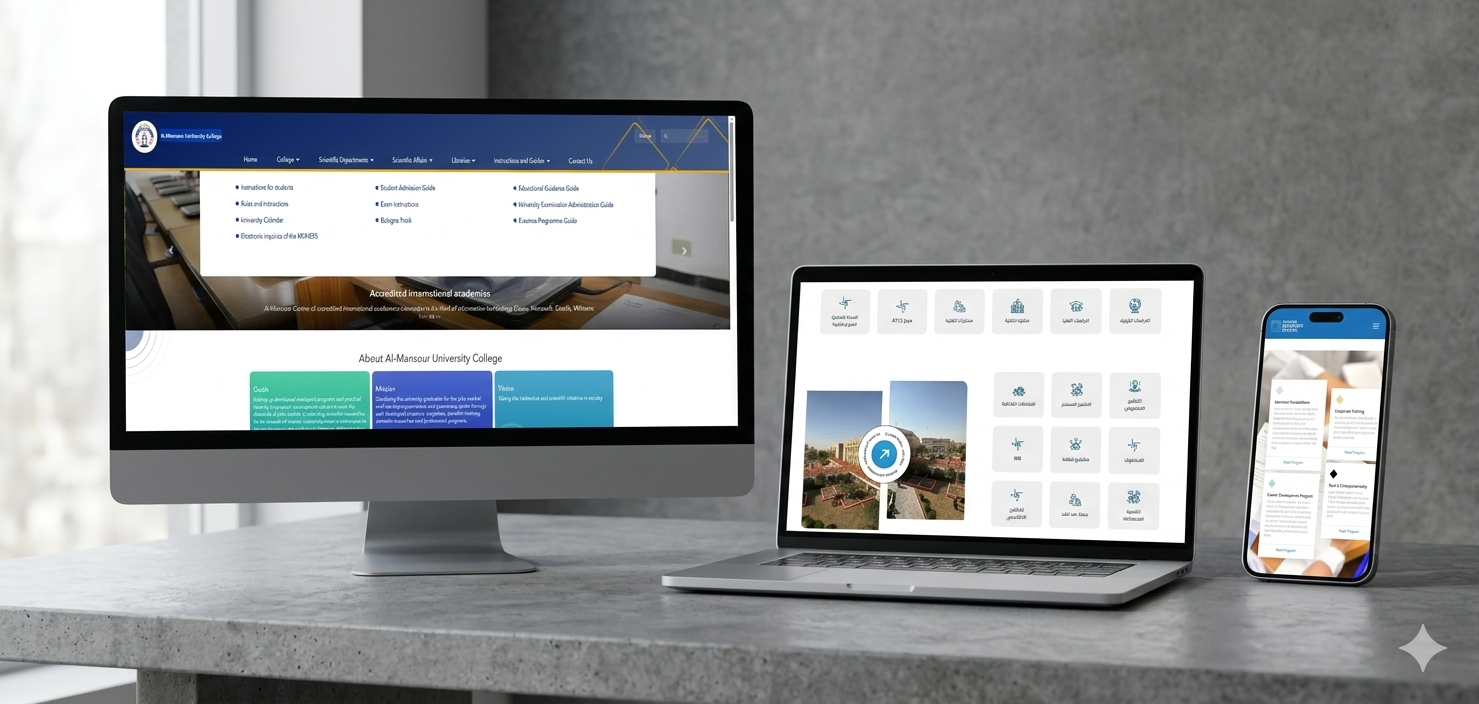
The NTP system was built from the ground up to address the specific logistical and medical requirements of the Iraqi healthcare landscape. Functioning as a comprehensive online stock management and patient monitoring platform, it enables the categorical observation of medications used to treat both MDR-TB and Drug-Susceptible TB (DS-TB). The system’s architecture follows the patient’s journey from the moment of registration at any of the country’s TB centers. By digitizing patient records, physician follow-ups, and pharmacy dispensations, the platform ensures that no individual falls through the cracks of a complex treatment regimen.
Beyond patient care, the system serves as a vital tool for inventory and quality control. It tracks the quantity of drug items, their sources, and essential quality control due diligence, ultimately producing detailed inventory reports. These reports are instrumental for health officials, providing the "actionable intelligence" required to oversee the flow of life-saving drugs across various governorates in real-time. Since becoming operational in 2022, the NTP system has moved beyond basic implementation into a phase of continuous evolution. Current development focuses on increasing accessibility through a mobile application and integrating a Directly Observed Treatment (DOT) form. This addition will allow surveyors to track medication adherence in real-time, directly addressing the challenge of patient non-adherence which often leads to further drug resistance.